|
Let stand for 2 minutes rinse with water as in step 3.ħ. Place the slide flat on the staining tray and dispense just enough Counterstain Solution (0.6% safranin in 20% ethanol) to cover each smear. Do this for 10 seconds and IMMEDIATELY rinse, as in step 3, with a gentle stream of water.Ħ. This step is tricky as it is easy to over- or under-decolorize. Lift the slide at a 45 degree angle and drip Decolorizing Reagent (80% isopropyl alcohol, 20% acetone) down the length of the slide making sure it comes in contact with all three smears. 
Let stand for 1 minute, and rinse thoroughly with a gentle stream of water as in Step 1.ĥ. Dispense just enough Gram's Iodine (mordant)to completely cover each smear. Rinse the slide by lifting it at a 45 degree angle (using gloves or a clothes pin or slide holder) in a very gentle stream of water that is directed above the top smear until the waste water coming off the bottom is relatively clear drain off excess water by touching the edge of the slide to a paper towel.Ĥ. Dispense just enough Crystal Violet solution (0.5% crystal violet, 12% ethanol, 0.1% phenol) to completely cover each smear and stain for 1 minute. If you do, it is much easier to prevent dye from running off the slide during the staining process and to be sure that your smears are evenly covered with each reagent.Ģ. It is important that the slide be level during staining so use paper towels under the tray to get it leveled. Be sure you evenly cover all the smears on the slide throughout this procedure.ġ. Prepare a bacterial smear slide (Find protocol at BISC209: Preparing a bacterial smear slide). Some organisms and dead or dying cells do not take up or lose the stain appropriately and can not be classified as either Gram positive or Gram negative.ġ. 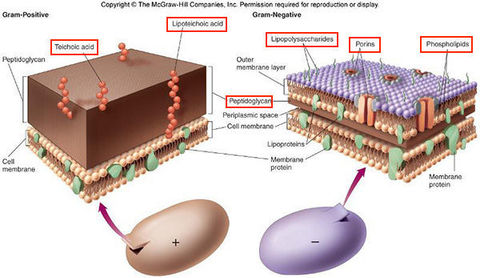
Purple (primary stain retaining) indicates Gram positive and red (counter stain uptake) indicates Gram negative. Due to the increase in porosity of the Gram negative cells after lipid loss from decolorization, safranin (counterstain) is able to permeate the cell wall of Gram-negative bacteria. The crystal violet-iodine complex remains within the Gram-positive bacteria because their cell walls lack the lipid-rich outer membrane of Gram-negative bacteria. The decolorizer extracts lipid from the cell wall of Gram-negative bacteria, so the crystal violet-iodine complex diffuses from these cells and loses color. Addition of the iodine leads to the formation of a crystal violet-iodine complex within the cell wall. Gram-positive and Gram-negative bacteria are both stained purple by the crystal violet (primary) stain. It is one of the most common and useful tools in identifying bacteria in the microbiology laboratory. This staining procedure is used to classify bacteria.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
